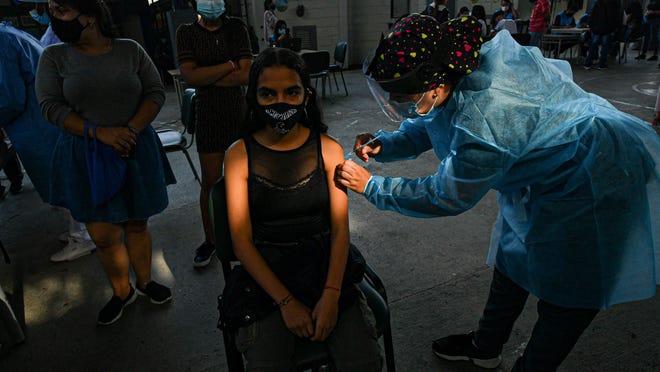BOSTON — Thomas Feldborg and Daria Rokina set off nearly every afternoon to explore this city. They leave from their hotel in the Charlestown neighborhood, pushing their baby carriage, some days heading deep into downtown, others choosing a path along the Charles River.
Every few minutes, Rokina stops to peek inside and check on 16-month-old Alissa. She gently rubs Alissa's cheek and coos a few soothing words, making sure the little girl is warm enough in her yellow snowsuit and adjusting her sparkly unicorn earmuffs.
Alissa rests better in the outside air. The daily walks allow the couple to relieve some of the stress of not knowing whether their daughter will survive – and if so, in what condition.
Feldborg, 50, has three older sons, and Rokina, 43, has one. Alissa, their first child together, was born Dec. 2, 2019, in Copenhagen, Denmark, where the family lives. For her first four months of life, Alissa seemed perfectly normal. She learned to roll and sit up. She babbled and grabbed for toys.
Then her progress began to stall. By six months when Alissa wasn't trying to crawl, Rokina took her to the pediatrician. Children advance at their own pace, the doctor assured her. Nothing was wrong.
At eight months, the trouble became impossible to ignore. Alissa was hospitalized, dehydrated and unable to eat. A neurological exam turned up cherry red spots at the back of her eyes, a devastating observation in a baby, because it indicates one of a few genetic disorders, most terrible and deadly before age 5.
16 month old Alissa gets gene therapy in a fight for her life.
16 month old Alissa Feldborg is undergoing gene therapy for Sandhoff disease, a very rare, fatal genetic disorder.
Robert Deutsch, USA TODAY
Feldborg immediately started Googling. Before Alissa received the diagnosis of Sandhoff disease, an extremely rare condition, he already had stumbled across the idea of gene therapy. If they could just repair the faulty gene Alissa inherited from him and Rokina, she might have a shot at life.
That internet research led the couple to the University of Massachusetts, where in late January, Alissa, then nearly 14 months old, became the first child in the world to receive a full dose of a new gene therapy. If it worked, it would trigger the cells in her brain to start making the enzyme they had been missing, clearing out cellular debris, so they can function normally.
Gene therapy like Alissa's – a scientific vision for decades – is finally becoming a more common reality in the United States.
Dozens of disorders are now being treated, though most still only in clinical trials, like Alissa's. Within the next few years, experts say, gene therapies could soon be available for conditions never effectively treatable before, such as sickle cell disease, Huntington's, ALS, Parkinson's, some forms of heart disease and a host of very rare diseases.
"The exponential growth phase" of gene therapy has arrived, said Dr. Cynthia Tifft, director of the Pediatric Undiagnosed Diseases Program at the National Institutes of Health.
"Finally, after literally decades of hearing it was just around the corner, we are witnessing some real successes," said Dr. Isaac Kohane, chair of the Department of Biomedical Informatics at Harvard Medical School and head of the federally funded Coordinating Center for the Undiagnosed Diseases Network.
At their best, gene therapies offer the possibility of a cure, truly reversing the root cause of a disease.
But as simple as the idea seems – just fixing a DNA typo – it is incredibly difficult to turn into an actual therapy. Scientists so far have spent decades on each step in the process.
And with such a new procedure, aimed at having lifelong benefits, it's too early to know whether these approaches will change a child's trajectory, allow an adult to live without fear and pain, or prevent a disease from ever coming back.
"People are underestimating what it's going to take to make (gene therapy) work in the short term," Kohane said, "and under-appreciating how transformative it will be in the long term."

On her first birthday, Dec. 2, 2020, Alissa received a colorful book and toy. She grabbed for both, excited to reach them.
But by the end of the month, she could no longer make that simple movement. She could barely hold up her head and slipped into silence. Seizures accelerated the regression.
"She went more into herself … she was not very present anymore," said Feldborg, who speaks near-perfect English.
He and Rokina felt totally alone deciding whether to pursue the treatment for Alissa. Two other babies had similar procedures with a lower gene therapy dose, but medical privacy laws prevented the couple from speaking with their parents.
There was no doubt what would happen if they did nothing.

For babies with Alissa's disorder and a "cousin" condition called Tay-Sachs disease, "their quality of life is essentially vegetative," Tifft said. Feeding tubes and other supportive care can keep them alive for a while. But they all die by the time they should be entering kindergarten.
So, as Feldborg said, the pair decided to "let hope conquer the fears." It was simply too awful to watch her decline without trying anything when they were among the first families in history who could try something.
But even as he and Rokina packed and boarded a plane for the U.S. in early January, they were tormented by the idea that the therapy might only keep Alissa alive longer in the same miserable state.
"Instead of having this cruel, short life of two, three, maybe four years, maybe she will just have a cruel, long life," Feldborg said, while walking through a park on a brisk, early spring afternoon. "That's what we were fearing."
On Jan. 26, after weeks of testing to make sure she was still a good candidate and prepare her for the procedure, Alissa was wheeled into an operating room at UMass Memorial Medical Center in nearby Worcester for brain surgery.
Researchers felt that the best way to unclog her brain cells was to inject the gene therapy directly into her thalamus, two oval structures deep inside the brain that act as sort of a highway junction, connecting paths from many parts of the brain. Doctors used an image-guided robotic arm to ensure it got to the right spot.
The next day, the same gene therapy was infused into the fluid around her spinal cord so it could penetrate the nerves there.
If it worked as planned, the added genes would instruct cells throughout her nervous system and brain to start producing the missing enzyme.
"You don't actually need a lot of enzyme activity in order for the neurons to function better," said Dr. Pavan Cheruvu, CEO of Sio Gene Therapies, the New York-based biotech company running the trial and paying the full cost of Alissa's treatment and her parents' stay in the U.S.
One previous child had received the thalamic surgery, but there wasn't enough of the therapy available to give her a full dose. So far, her condition has been stable at a time when most kids with the condition decline, said Dr. Terence Flotte, dean of the UMass Medical School, who is leading Alissa's trial. Another child got the spinal injection but not the thalamic one.
Alissa was the first to get a full dose in both places. Later patients in the trial will get an even higher dose. Feldborg and Rokina hope Alissa got enough to make a profound and positive difference in the course of her life.
But for now, all they can do is wait. And walk.


About 45 minutes west of Boston, a herd of Jacob sheep live in a series of pens overlooking rolling hills.
Guarded by a llama and an electric fence, both to keep coyotes away, several sheep pause to stare at strangers at the Cummings School of Veterinary Medicine at Tufts University where they live. Then they bolt off.
These 110 sheep are the descendants of a pair that once lived on a Texas farm. In 1999, two lambs began stumbling and tripping. They developed seizures, lost sight and had trouble swallowing before dying young. Owners Fred and Joan Horak wanted to understand why.
It took a decade for a researcher from New York University Medical Center, now NYU Langone Health, to figure out the sheep carried the same genetic mutation that causes Tay-Sachs disease. Researchers later learned cats also can inherit a similar genetic mutation.
Finding an animal model in which a disease naturally occurs is extremely helpful for developing treatments, said Douglas Martin, a professor at the Auburn University College of Veterinary Medicine in Alabama who studies these conditions in house cats.
In animals, researchers tested and refined the virus that eventually delivered Alissa's gene therapy.

Gene therapies are often transported into cells by viruses – similar to how pathogens are used to deliver the COVID-19 vaccines made by Johnson & Johnson and AstraZeneca-Oxford University. It's taken decades to find the appropriate viruses and engineer them to safely deliver genes or editing tools.
In Alissa's trial, a virus carries DNA instructions for making the missing enzyme. Designed by Miguel Sena-Esteves, a UMass Medical School researcher who has been working on the project for more than a decade, the therapy delivers two genes, even though Alissa is only missing one. Animal studies showed that adding both the gene that causes Tay-Sachs plus a nearby one that causes Sandhoff provides the best results for children with both conditions.
Alissa's own DNA isn't changed in this approach, though other gene therapies rely on gene editing to alter cells' DNA code.
Her immune system is kept tamped down with medication so that, if needed, she can be dosed again with the virus-carrying gene therapy, Flotte said.

Researchers have tried before to treat both conditions by providing the missing enzyme instead of adding genes, but it is too big to cross from the bloodstream into the brain.
By 2012, Sena-Esteves had shown that the virus and gene therapy worked in mice; Martin, at Auburn, proved the same in cats, and a third colleague, Heather Gray-Edwards, now of UMass Medical School, used it to rescue Jacob sheep.
Just before trying the approach in children, the team decided to test it in monkeys. The Food and Drug Administration probably wouldn't have required it after their success in other animals, but they wanted to be extra cautious.
The results were devastating. The monkeys became apathetic and lost dexterity, Edwards said. They clearly weren't helped.
It took years of research to figure out that the animals had gotten too much of a good thing: The extra enzymes that helped clean up the brain cells of other mammals was overwhelming and killing the monkey's cells.
Sena-Esteves said the day he learned the results was perhaps the worst of his professional life. But it was far better to learn the lesson on monkeys.
The therapy given to children is carefully calibrated in hopes of achieving a Goldilocks balance: not too little, but not too much.


Last year, Feldborg and Rokina, both now on paid leave from their sales and logistics jobs, befriended the other two Danish families whose babies had been diagnosed with Sandhoff.
One child was two months older than Alissa, the other eight months older. Neither got gene therapy. Both died earlier this year.
Doctors selected Alissa for the trial hoping she was still young enough and her losses recent enough that the damage to her brain might be reversible.
"We think that there are many cells that are simply under duress" due to the toxic build-up of fats, said Dr. Florian Eichler, a pediatric neurologist at Massachusetts General Hospital who treats Alissa. "If we can appropriately return a healthy copy of the gene and the enzyme, that cell can recover."
Last fall, watching their daughter slip further away, Feldborg and Rokina worried that Alissa would be disqualified from the trial because she had already lost too much function, or that the treatment would come too late to make a difference.

Doctors warned them not to expect much. No one knew if the therapy would help or how long it might take to start seeing changes.
But just a week after the surgeries, Alissa's sparkling blue eyes, which had been rolling randomly and constantly, seemed to stabilize and focus. With eyes in constant motion, she couldn't have been able to see much.
Now that they're more focused, it's also easier to feel like there's someone present behind them.
In February, Alissa started to move her hands with some intention and began eating more. Swallowing was the one skill she hadn't lost, but perhaps because of the steroids that are part of her care she became hungry again, accepting spoonfuls of soft food. Recently, she learned how to suck down a bottle for the first time.
"If this development will continue, we are just so very happy," said Rokina, who is not as comfortable speaking English as her husband. "It's so nice."
There also was a wonderful surprise. About two weeks after her surgeries, Alissa started smiling again. Not in response to the outside world, but to some internal moment of pleasure or humor.
A few days after that, she laughed a deep, guttural belly laugh. Feldborg said he can count on one hand the number of times she had laughed like that before the procedure. But for the last few weeks, she has enjoyed a private laugh nearly every day.
The sound of those belly laughs has been a gift, a balm.
The first time it happened, while giving her a bath, Feldborg said, "We were so happy, we just kissed each other like we had become world champions in our favorite sports."

To compensate for her lack of sight and hearing, Rokina and Feldborg try to offer Alissa some connection to the world through touch.
Rokina, a native of Russia, grew up with the tradition of giving babies massages. Before Alissa's gene therapy, months of massages and movement therapy had done little except perhaps allow her to keep swallowing.
After the surgeries, Feldborg and Rokina renewed the practice, driving Alissa once a week to baby massage sessions in a far-flung Boston suburb, and two or three mornings a week to the closer-in town of Arlington, where Matty Wilkinson tries to reconnect the little girl's brain to the body she can do so little with.
Wilkinson, who has a master's degree in child development as well as training in the Feldenkrais Method and the Anat Baniel Method for Children, uses small movements aimed at teaching Alissa's brain to once again sense her neck, arms, legs, pelvis and spine.

"She's having to rebuild a map of herself, how she can interact with her environment," he said after a recent session. "I'm trying to make the information clearer and more distinct, so she can perceive those feelings … so she can gain more choice."
Laying Alissa on her side, Wilkinson pulls gently on her left arm while touching the back of the same shoulder. She occasionally moves her mouth in a sucking motion, but otherwise lies still.
One of the first truly successful gene therapies, for a condition called spinal muscular atrophy, showed that the treatment, while immensely effective, wasn't enough on its own. Just as newborns learn to control their limbs by moving them, so children whose brains have lost the ability to direct movement – or never developed it in the first place – need practice for the proper wiring to form.
"We say with the nervous system, you have to use it or you lose it," Eichler said. "There has to be continued stimulation, activity, and so all of those things Alissa's parents do so well with her are vital."

People tell Feldborg how "brave" he and his wife are for trying gene therapy with Alissa. He's not sure how to respond.
"There's a fine line between bravery and stupidity," he said. "Are we so stupid in our hope? Is it a fool's mission we're on?"
This past week, three months after her surgery, doctors began a battery of tests on Alissa to see if they can detect objective signs of progress. They're hoping to see evidence on her MRI scan that her brain is repairing itself.
The trial would be deemed successful if it achieved any of three possible outcomes, said the NIH's Tifft: It could slow Alissa's decline, keep her from slipping any further or actually help her gain skills.
"None of these children ever improve on their own. They just don't," Tifft said. "In a disease that only progresses downhill, even preserving function is a win."
"Then the question is, for how long," she added.
No matter what happens, the trial represents "a huge milestone" in the treatment of Sandhoff and related diseases, Eichler said.
Tay-Sachs was identified more than a century ago. The chemistry of what it and Sandhoff do to the brain has been understood for at least half that time. Now, the gene deficit itself can finally be addressed, Eichler said.
Still, a lot to be learned about the best timing for the therapy, what cells need to be targeted and what meaningful change looks like.
"That is the nature of progress. If we knew what works, we wouldn't need to conduct trials," Eichler said. "I feel for the parents."
For their part, Feldborg and Rokina say they hope the trial will help others, but are realistic about what might be achievable for themselves. They know Alissa will never be like other children. They just want a daughter they can communicate with in some way.
"She can be in a wheelchair or whatever," Feldborg said, "but if we don't get a thinking person out of this, that's where I think we may have been more stupid than brave."
Contact Karen Weintraub at [email protected].
Health and patient safety coverage at USA TODAY is made possible in part by a grant from the Masimo Foundation for Ethics, Innovation and Competition in Healthcare. The Masimo Foundation does not provide editorial input.
Source link